Sodium thiosulfate, the stable sodium salt for thiosulfuric acids, is instabile in its free state. Sodium thiosulfate is a synthetic compound known in chemistry as sodium sulphate, and in the food industry as additive E539, approved for use in food production. Sodium thiosulfate is used in gold mining, water treatment, analytical chemistry, the development of silver-based photographic film and prints, and medicine. It is on the World Health Organization’s List of Essential Medicines. The medical uses of sodium thiosulfate include treatment of cyanide poisoning and pityriasis.
Characteristics
Thiosulfate is an ion with acid-base, reactive and ligand properties. Endothermic reaction occurs when sodium thiosulfate is dissolved in water. This means that the solvent’s temperature decreases. It melts at 56 degrees Celsius in crystallization water, and it is lost completely at 100 degrees Celsius; it also decomposes at higher temperatures. It is reductive because acids decompose the solutions.

| System Name | Sodium thiosulfate |
|---|---|
| Traditional Naming | Na2S2O3 |
| International Marking | E539 |
| Traditional Naming | Sodium sulphate, soda hyposulfite (sodium), antichlorine |
| Group | Inorganic thiosulfates (salts) |
| Aggregate state | Colorless monoclinic crystals (powder) |
| Solubility | Soluble in water, insoluble in alcohols |
| Critical temperature | 220°C |
| Melting Point | 50 °C |
| Properties | Reducing (antioxidant), complexing |
| Dietary Supplement Category | Acidity regulators, anti-caking agents |
| Toxicity | Not tested, conditionally safe |
| Origin | Synthetic |
| Applications | Food, textile, leather, photographic, pharmaceutical, analytical chemistry |
Cod CAS
10102-17-7
Pharmacological action
Pharmacological action: detoxifying, desensitizing and anti-inflammatory.
Pharmacology
It forms non-toxic and low-toxic compounds when combined with heavy metals, salts of halogens, and cyanides. It has antidote qualities in relation to aniline and benzene, copper, mercury hydrocyanic acid, chloride, phenols, and iodine. Nonpoisonous sulfurites can be formed when poisoned with arsenic or mercury, and lead compounds. The main mechanism of detoxification in poisoning by hydrocyanic acid and its salts is the conversion of cyanide into thiocyanate ion, which is relatively non-toxic, with the participation of the enzyme rodonase – thiosulfate cyanide-seratransferase (found in many tissues, but maximum activity is shown in the liver). Although the body is capable of detoxifying cyanides, the rhodonase enzyme system works slowly and its activity in cyanide poisoning is not sufficient. To accelerate the reaction catalyzed rhodonase causes, it is necessary for exogenous sulfur donors to be introduced, most commonly sodium thiosulfate.
It is used to detoxify patients suffering from alcoholic delirium. After IV administration, sodium Thiosulfate can be distributed in extracellular fluid and excreted unchanged from the urine. The biological T1/2 is 0.65 hours.
The anti-scabies activity of the sulfur dioxide and sulfur in the environment it decomposes in is due to their ability to produce sulfur and sulfur dioxide. These substances have a detrimental effect on the eggs and the scabies mite. Non-toxic sodium thiosulfate Studies on dogs receiving continuous sodium thiosulfate infusions showed hypovolemia. This is likely due to the osmotic diuretic effects of sodium thiosulfate.
Graphic (structural) formula for sodium thiosulfate
The structural (graphical) formula for sodium thiosulfate is more obvious. It shows how the atoms are connected within the molecule:

Chemical formula of sodium thiosulfate
Chemical formula of sodium thiosulfate Na2S2O3. It shows that this molecule contains two sodium atoms (Ar = 23 a.m.u.), two sulfur atoms (Ar = 32 a.m.u.) and three oxygen atoms (Ar = 16 a.u. .m.). According to the chemical formula, you can calculate the molecular weight of sodium thiosulfate:
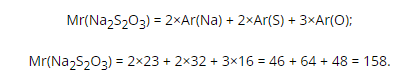
Ionic formula
Sodium thiosulfate is an electrolyte that dissociates into ions in aqueous solution according to the following reaction equation:

Precautionary measures
The reagent is fire and explosion-proof, slightly toxic. It acts as an irritant on contact with the skin and mucous membranes of the eyes and respiratory organs. Store sodium thiosulfate pentahydrate in dry, well-ventilated areas.
Risks and dangers
At 45ºC it decomposes on exposure to heat and may react violently in the presence of nitrites and peroxides , creating an explosion hazard . In the presence of acids, the formation of hazardous sulfur oxides can occur.
Application
- for cleansing tissues from iodine;
- for removing traces of chlorine after bleaching fabrics;
- reagent in iodometry;
- registered in the food industry as food additive E539;
- antidote for poisoning: As, Br, Hg and other heavy metals, cyanides (translates them into thiocyanates), etc;
- fixer in photography;
- for intestinal disinfection;
- for the extraction of silver from ores;
- for the treatment of scabies (together with hydrochloric acid);
- can be used as a medium for determining molecular weights by freezing point depression (cryoscopic constant 4.26°);
- anti-inflammatory and anti-burn agent;
- additives for concrete.




